  Since entropy is a state variable, just depending upon the beginning and end states, these expressions can be used for any two points that can be put on one of the standard graphs. To illustrate this concept, the equation relating free energy change to the enthalpy and entropy changes for the process is considered: G H T S G H T S The spontaneity of a process, as reflected in the arithmetic sign of its free energy change, is then determined by the signs of the enthalpy and entropy changes and. pressure 100 kPa, 20 degrees C or 1 atm and 0 degrees C, etc.) - not absolute entropy, down to the solidus or liquidus. Using the ideal gas lawīut since specific heats are related by C P = C V + R. begingroup Im not familiar with this software, but I would say that likely your 'Entropy' function is actually giving you an entropy change, presumably from some 'standard' conditions (e.g. 
This is a useful calculation form if the temperatures and volumes are known, but if you are working on a PV diagram it is preferable to have it expressed in those terms. Making use of the first law of thermodynamics and the nature of system work, this can be written Entropy is the measure of the disorder of a system and can is the energy of a system over its temperature, represented as J/K. Thus, entropy measurement is a way of distinguishing the past from. entropy converts any class other than logical to uint8 for the histogram count calculation so that the pixel values are discrete and directly correspond to a. As one goes 'forward' in time, the second law of thermodynamics says, the entropy of an isolated system can increase, but not decrease. With kT/2 of energy for each degree of freedom for each atom.įor processes with an ideal gas, the change in entropy can be calculated from the relationship Entropy is one of the few quantities in the physical sciences that require a particular direction for time, sometimes called an arrow of time. , F (1) 1, F (2) 1 and this formula has a much lower entropy and applies to any length of the Fibonacci sequence. This gives an expression for internal energy that is consistent with equipartition of energy. The first 128 symbols of the Fibonacci sequence has an entropy of approximately 7 bits/symbol, but the sequence can be expressed using a formula F (n) F (n1) + F (n2) for n 3, 4, 5. Then making use of the definition of temperature in terms of entropy: Allowing to change the number of microstates without affecting the total energy of the system is in discrepancy with equation 1 and 2. 3Nh2 32 + 5 from the microcanonical (NV E) ensemble. Expanding the entropy expression for V f and V i with log combination rules leads toįor determining other functions, it is useful to expand the entropy expression using the logarithm of products to separate the U and V dependence. One of the things which can be determined directly from this equation is the change in entropy during an isothermal expansion where N and U are constant (implying Q=W). 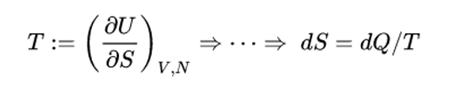
The entropy S of a monoatomic ideal gas can be expressed in a famous equation called the Sackur-Tetrode equation. Although this result was obtained for a particular case, its validity can be shown to be far more general: There is no net change in the entropy of a system undergoing any complete reversible cyclic process.Entropy of an Ideal Gas Entropy of an Ideal Gas There is no net change in the entropy of the Carnot engine over a complete cycle. 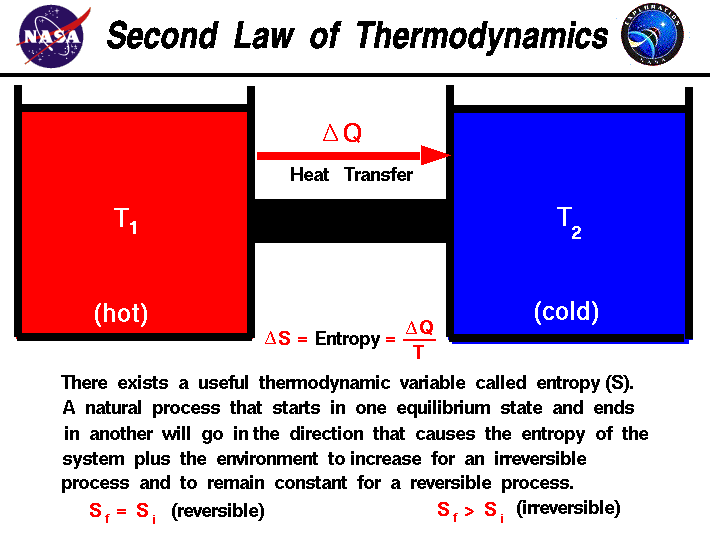
However, we know that for a Carnot engine, First it's helpful to properly define entropy, which is a measurement of how dispersed matter and energy are in a certain region at a particular. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
